Kūpeke Water Quality Measurements and Maps
Water quality data collected: August 2021 Measured using: Eureka Manta+ 35 Water Probe
Measurements taken: Temperature, pH, Conductivity, Turbidity, Dissolved Oxygen (DO), Chlorophyll, and Blue Green Algae
Maps Created By: Richard Gill Laboratory - Brigham Young University Provo Contact: Kalaʻi [email protected]
Data Collection Sites
This map shows the location of where water quality measurements were taken within the pond. They represent a single measurement in time and are not a good indicator of average conditions as water quality rapidly changes depending on the surrounding conditions.
This map shows the location of where water quality measurements were taken within the pond. They represent a single measurement in time and are not a good indicator of average conditions as water quality rapidly changes depending on the surrounding conditions.
Blue Green Algae
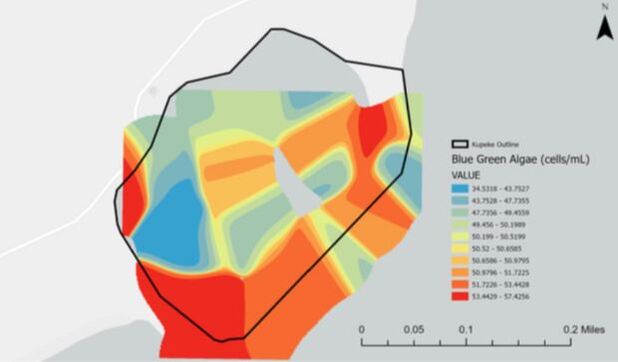
Blue green algal blooms can pose a threat to many ecosystems as their cyanobacteria can produce a variety of different toxins. Many of the toxins can have a detrimental effect on fish and other marine life populations, and that is why preventing the algal blooms from growing and spreading is so important. Additionally, blue green algae can cause oxygen levels of the surrounding water to drop which can dramatically affect the environment for fish and other animals within the ecosystem. From the map we are able to see areas with concentrated levels of blue green algae which helps us to identify areas of potential concern.
Blue green algal blooms can pose a threat to many ecosystems as their cyanobacteria can produce a variety of different toxins. Many of the toxins can have a detrimental effect on fish and other marine life populations, and that is why preventing the algal blooms from growing and spreading is so important. Additionally, blue green algae can cause oxygen levels of the surrounding water to drop which can dramatically affect the environment for fish and other animals within the ecosystem. From the map we are able to see areas with concentrated levels of blue green algae which helps us to identify areas of potential concern.
Chlorophyll
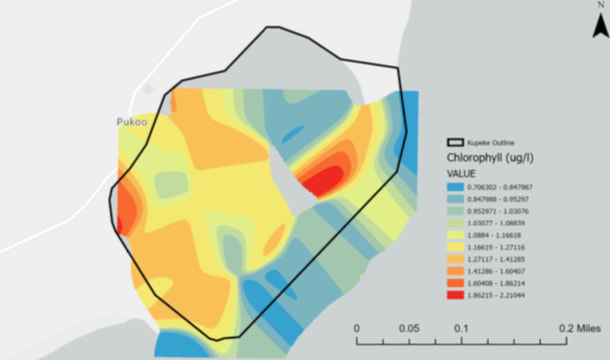
Chlorophyll, an essential component in photosynthesis, can also serve as an indicator of the presence of potentially harmful algal blooms. Areas with high concentrations of chlorophyll can help us identify areas to monitor to ensure the water is at its prime and remains free of toxic algal blooms. The map indicates areas with higher chlorophyll which might be prone to toxic algal development. By carefully monitoring the area, precautions can be taken to prevent the spread of toxic blue green algae and other harmful components.
Chlorophyll, an essential component in photosynthesis, can also serve as an indicator of the presence of potentially harmful algal blooms. Areas with high concentrations of chlorophyll can help us identify areas to monitor to ensure the water is at its prime and remains free of toxic algal blooms. The map indicates areas with higher chlorophyll which might be prone to toxic algal development. By carefully monitoring the area, precautions can be taken to prevent the spread of toxic blue green algae and other harmful components.
Conductivity
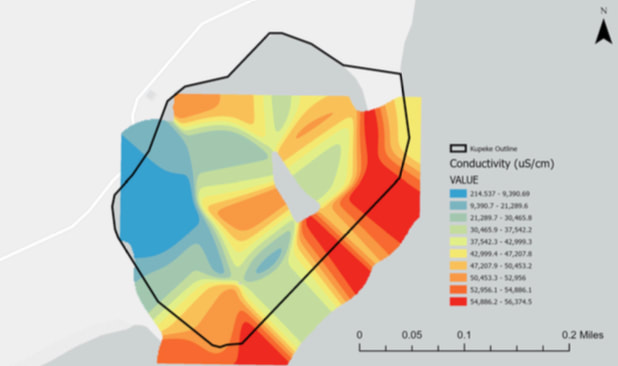
Conductivity is the measure of water’s ability to pass an electrical current and is influenced by the concentration of dissolved ions in the water. The higher the concentration, the higher the conductivity. Conductivity is also affected by water temperature, increasing in warmer water and decreasing in cooler water. Industrial, agricultural, and human wastewaters, as well as polluted runoff, can increase water conductivity as they carry additional chloride, phosphate, and nitrate ions into the fishpond. High levels of these conductive ions can be harmful to marine life. Freshwater springs can decrease the conductivity of seawater by adding water low in dissolved ions. As an increased concentration of ions often is correlated with increased salts in the water, conductivity and salinity are related. Conductivity is an indirect measure of salinity and the effects of varying conductivity levels on marine life can resemble the effects of varying salinity.
Conductivity is the measure of water’s ability to pass an electrical current and is influenced by the concentration of dissolved ions in the water. The higher the concentration, the higher the conductivity. Conductivity is also affected by water temperature, increasing in warmer water and decreasing in cooler water. Industrial, agricultural, and human wastewaters, as well as polluted runoff, can increase water conductivity as they carry additional chloride, phosphate, and nitrate ions into the fishpond. High levels of these conductive ions can be harmful to marine life. Freshwater springs can decrease the conductivity of seawater by adding water low in dissolved ions. As an increased concentration of ions often is correlated with increased salts in the water, conductivity and salinity are related. Conductivity is an indirect measure of salinity and the effects of varying conductivity levels on marine life can resemble the effects of varying salinity.
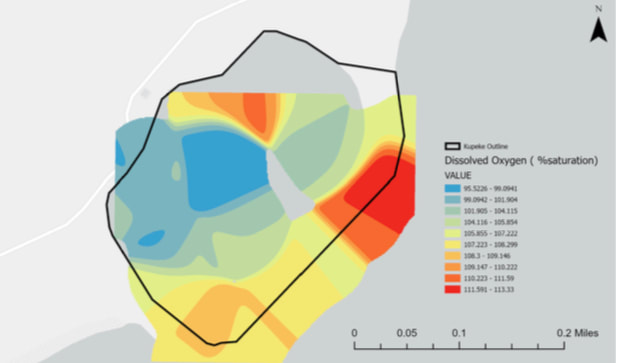
Dissolved Oxygen
Most bottom dwelling or bottom feeding fish typically need dissolved oxygen concentrations of about 1-6 mg/L, and for most shallow water fish the concentration levels range from 4-15 mg/L. Based off of this map we can see that the current range of dissolved oxygen levels would be more accommodating to bottom feeding fish, but shallow water fish would still probably be able to do just fine. The dissolved oxygen levels can be impacted by changes in water temperature, salinity, as well as atmospheric pressure. With changes in any of these factors we can expect the animal life to be impacted as well.
Most bottom dwelling or bottom feeding fish typically need dissolved oxygen concentrations of about 1-6 mg/L, and for most shallow water fish the concentration levels range from 4-15 mg/L. Based off of this map we can see that the current range of dissolved oxygen levels would be more accommodating to bottom feeding fish, but shallow water fish would still probably be able to do just fine. The dissolved oxygen levels can be impacted by changes in water temperature, salinity, as well as atmospheric pressure. With changes in any of these factors we can expect the animal life to be impacted as well.
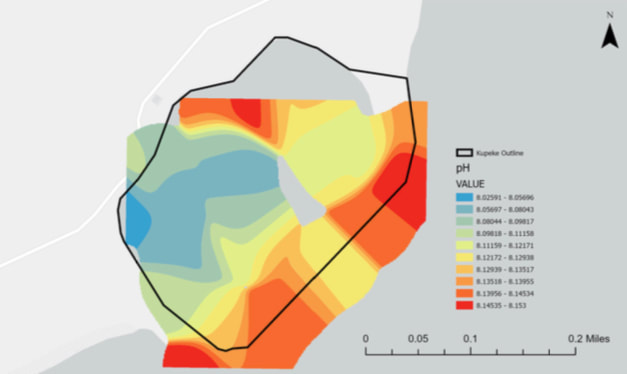
pH
The pH of seawater is important because it affects the chemical reactions that occur within it, as well as equilibrium conditions and toxicity. Factors that affect pH levels are runoff from precipitation, rivers and streams feeding into the ocean, and ocean currents.
When pH is low, organisms that have calcium carbonate shells and skeletons struggle to survive. Acidic water can eat away at the existing calcium carbonate structures, as well as make it difficult to grow new ones. Even organisms that don’t have shells are affected by acidic waters. If the pH of water lowers, fishes’ bodies also become more acidic slowing their growth and inhibiting their ability to sense their surroundings.
The pH of seawater is important because it affects the chemical reactions that occur within it, as well as equilibrium conditions and toxicity. Factors that affect pH levels are runoff from precipitation, rivers and streams feeding into the ocean, and ocean currents.
When pH is low, organisms that have calcium carbonate shells and skeletons struggle to survive. Acidic water can eat away at the existing calcium carbonate structures, as well as make it difficult to grow new ones. Even organisms that don’t have shells are affected by acidic waters. If the pH of water lowers, fishes’ bodies also become more acidic slowing their growth and inhibiting their ability to sense their surroundings.
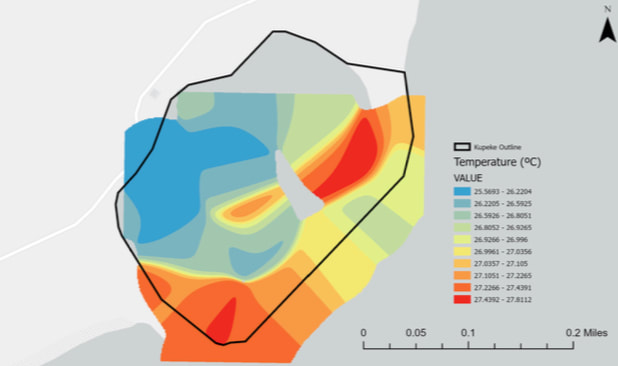
Temperature
The temperature of ocean water varies due to differences in water depth, latitude, and weather. Additionally, most of the heat produced by greenhouse gas-caused global warming is absorbed by the oceans. Increasing temperatures lead to coral bleaching as the heat stresses coral polyps, causing them to expel their symbiotic algae. Higher temperatures can lead to a loss of breeding grounds, cause migrations, and harm ocean ecosystems. Warmer water is also able to hold less dissolved oxygen when compared to cooler water. Single point temperature measurements are not a good representation of average temperature as fluctuations in surface temperature varies greatly throughout the day.
The temperature of ocean water varies due to differences in water depth, latitude, and weather. Additionally, most of the heat produced by greenhouse gas-caused global warming is absorbed by the oceans. Increasing temperatures lead to coral bleaching as the heat stresses coral polyps, causing them to expel their symbiotic algae. Higher temperatures can lead to a loss of breeding grounds, cause migrations, and harm ocean ecosystems. Warmer water is also able to hold less dissolved oxygen when compared to cooler water. Single point temperature measurements are not a good representation of average temperature as fluctuations in surface temperature varies greatly throughout the day.
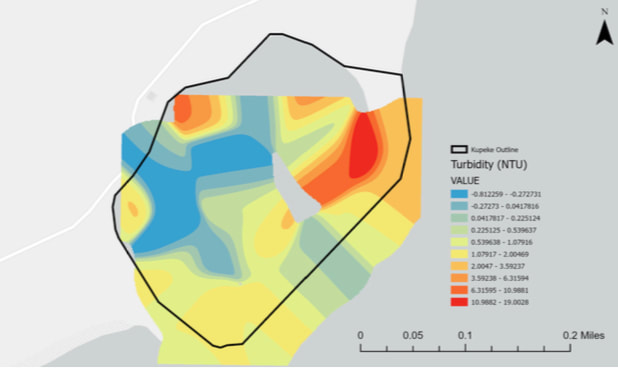
Turbidity
Turbidity is the measure of clarity in water, how much light is reflected and passed through the water. Silt, clay, pollution, algae, and other matter are what affect the levels of turbidity. In this graph, the higher levels of turbidity indicate the more unclear or murky that the water is, lower levels indicate that the water is clear.
Turbidity is the measure of clarity in water, how much light is reflected and passed through the water. Silt, clay, pollution, algae, and other matter are what affect the levels of turbidity. In this graph, the higher levels of turbidity indicate the more unclear or murky that the water is, lower levels indicate that the water is clear.